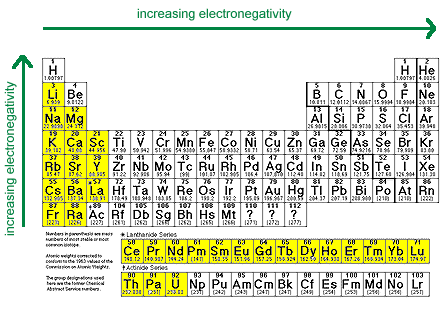
When an ion keeps the same charge in a reaction and is just there to balance out the opposite charges, we call it a Spectator Ion. Nitrate ion in both the reactants and the products has a -1 charge. In the equation below, nitrate is there to balance the charges. Positive ions (cations) can't just be floating around by themselves in solution, they have to have a negative ion (anion) to balance them. We say that Zn is more active than the copper. Metallurgists also apply the same principle during the extraction of metals.\): An activity series for common metals.Įxample: Zn and Cu both tend to lose 2 electrons, but the zinc wants to lose them more, so much so that it can force the two electrons on Cu 2 + and make it become Cu again. Why is knowledge of the activity series for metals important?Īctivity series of metals is essential in predicting the double displacement reactions. This series has metal atoms arranged from higher reactivity to lower reactivity. The activity series of metals describes the relative reactivity of metals against the standard hydrogen. Which series is the most reactive series? Therefore it will readily lose the electron. Moreover, the effective nuclear charge on that electron is the least due to the presence of five electronic shells between the nucleus and the electron.  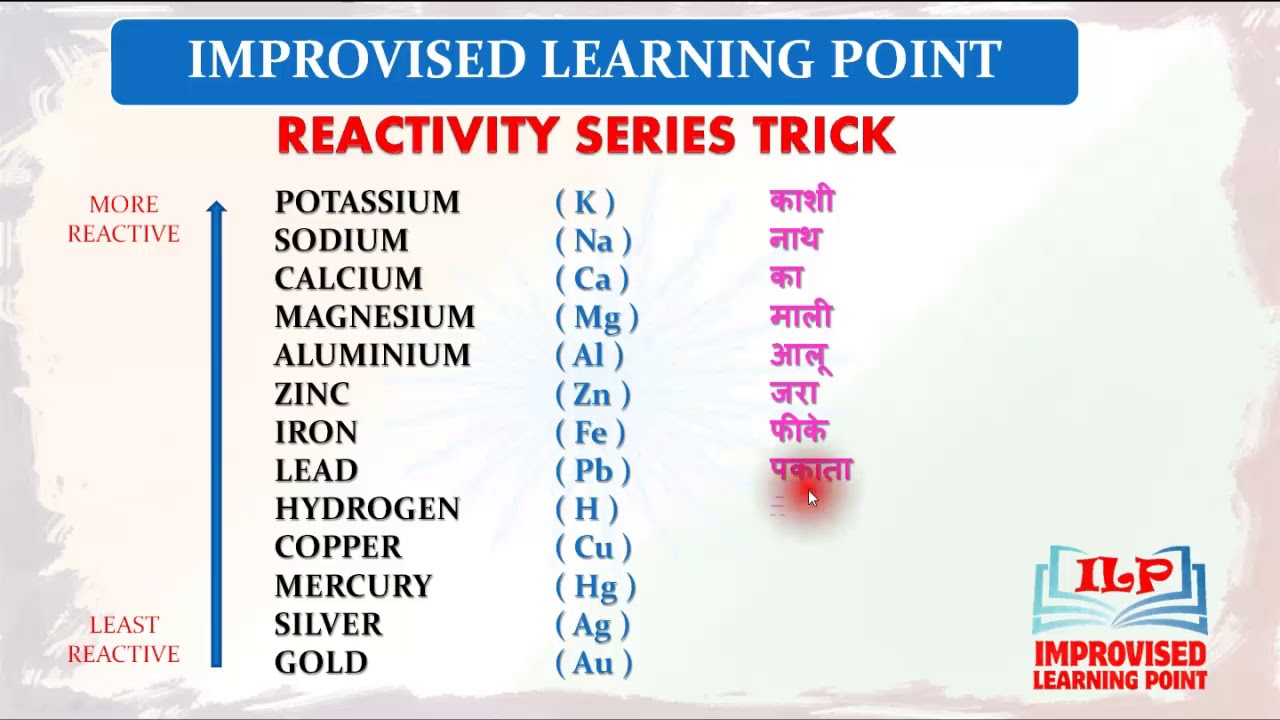
In this case, cesium has a single electron in its outermost shell. The reactivity of metal depends upon the oxidation potential (ability to lose electrons). Thus, zinc is more reactive.Ĭesium (Cs) is the most reactive metal in the periodic table. So, it is more susceptible to losing electrons than lead. Zinc (Zn) is present above in the activity series form lead (Pb). Key Point: Remember the common members of the activity series this way: Hydrogen gas is liberated when reactive metals come in contact with mineral acids. For example, reactive metals are added to the solution of less reactive metals for extraction purposes. Metals displacement reactionsĪctivity series of metals can be used to predict metal displacement. For example, at room temperature zinc does not react with water but above 50✬ it reacts, and hydrogen bubbles are witnessed. On going down the activity series, the tendency of metals to react decreases. The topmost metals react vigorously with water to liberate hydrogen gas. The reaction of metals with water can be predicted with activity series of metals. It also explains why aluminum forms a protective (insoluble) layer of aluminum oxide (Al 2O 3) and seems unreactive.Īpplications and uses of activity series of metals 1.Less reactive metals will not react even with hot water. 
Highly reactive metals can react with cold water.Hydrogen cannot reduce oxides of metals present above it in the series.Everything above hydrogen in the list can displace hydrogen from its acid.Some prominent outcomes of the activity series are given below: The activity series places metals in order of their tendency to participate in chemical reactions. The significant features of the activity series Therefore it can replace the copper from the CuSo 4 solution and become Zn 2+. This means that the metals with more than one electron in the outermost shell are more reactive than the ones having just one electron.įor example, zinc is more reactive than copper. The reason is the additional shell makes farther the leaving electron. The activity of alkali metals increases down the group. Alkali metals have one outermost electron so they can lose their electron to gain a stable configuration. Some metals are more reactive than others because of their electronic configuration. The activity of metals is defined by their ability to lose electrons and become ions. Moderate reactive metal, React with acids Highly reactive metals, even react with water It is important to note that this activity series is not absolute and can vary slightly depending on the specific conditions of the reaction. Moreover, this activity series describes the loss of electrons (oxidation potential), corrosive nature, and energy required for metal extraction processes from the ores. The more reactive metal will displace the less reactive metal from a compound. For example, if a metal higher in the activity series reacts with a compound containing a metal lower in the series, a single displacement reaction will occur. The activity series can help predict if a single displacement reaction will occur. The more reactive a metal is, the more likely it is to lose electrons and form a positive ion. The reactivity of these metals is usually determined by their ability to form positive ions. In the series, elements on the top are more reactive than those at the bottom. Activity series or reactivity series of metals explain certain properties of metals, mainly the reactivity of metals.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
